
Chief scientist on Operation Warp Speed resigns
The scientific adviser who guided U.S. efforts to speed up development of vaccines for Covid-19 is resigning at the behest of the incoming Biden administration.

The scientific adviser who guided U.S. efforts to speed up development of vaccines for Covid-19 is resigning at the behest of the incoming Biden administration.
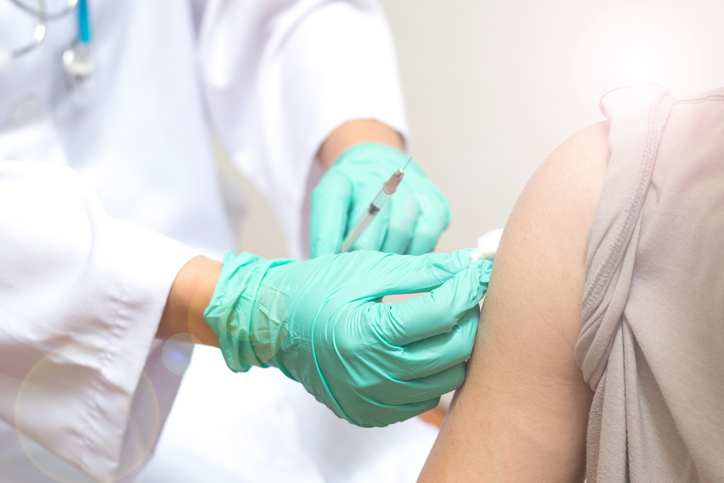
The CEOs of Pfizer, Moderna, McKesson and CVS Health explained that the distribution effort has gone well but the administration of the vaccine has now to catch up. They expect it to happen soon.

CVS Health’s infusion business will participate in a pilot to administer a recently authorized monoclonal antibody treatment to prevent patients from getting seriously ill with Covid-19. But data about the treatment, bamlanivimab, is mixed, and supply is limited.

The companies will supply 100 million doses of the vaccine by the end of the year, and the government will have the option to acquire 500 million more. However, the FDA must grant an authorization based on its safety and efficacy, for which there are only preliminary data so far.

The NIAID create the Covid-19 Prevention Trials Network by combining four existing ones, primarily focused on HIV and AIDS, which will continue that work in addition to their new focus on Covid-19.
News media reported that Moncef Slaoui would divest his shares in Moderna, which netted him at least $3 million after the company reported promising preliminary data on its Covid-19 vaccine Monday.

At a time when AI is reshaping pharma, Reverba Global CEO Cheryl Lubbert explained in an interview why empathy, context, and ethics still require a human touch.